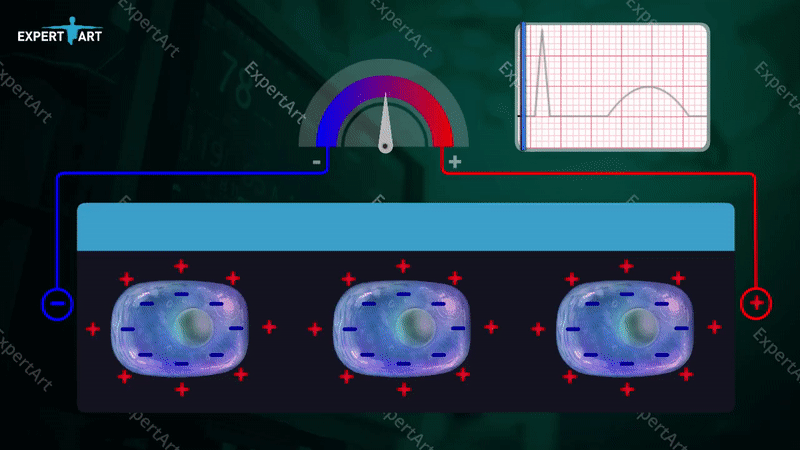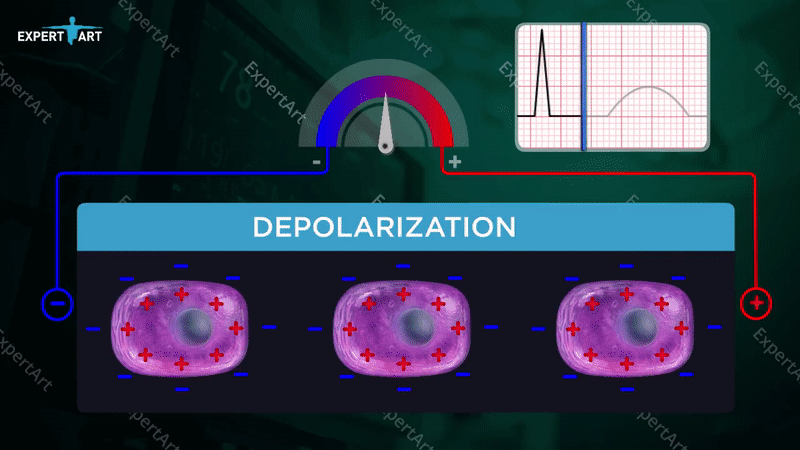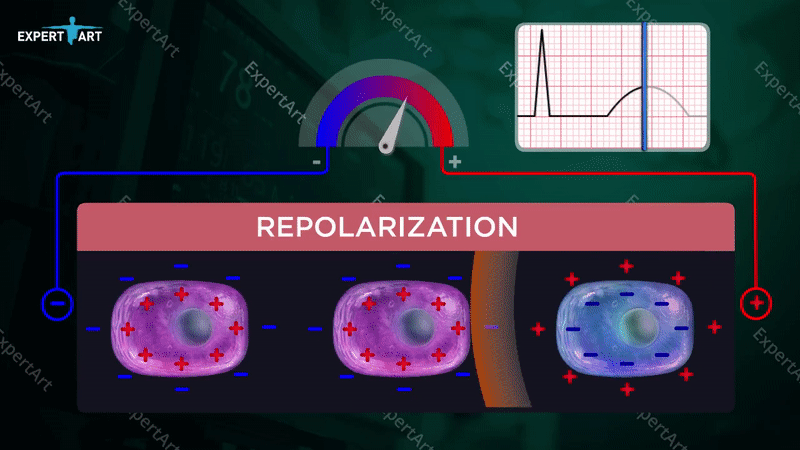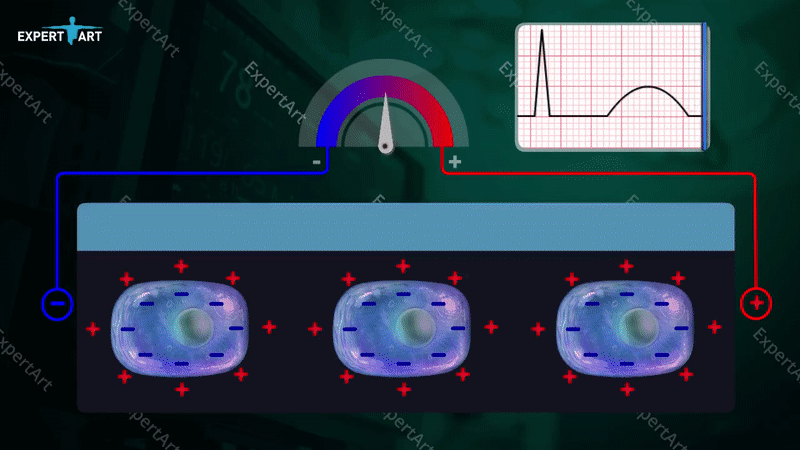
The bicarbonate buffer system is used to maintain the pH in the blood, which is necessary to maintain proper metabolic function. This system involves:
- Carbonic acid (H2CO3)
- Bicarbonate ion (HCO3-)
- Carbon dioxide (CO2)
Carbon dioxide (CO2) reacts with water (H2O) and forms carbonic acid (H2CO3). This acid quickly separates to form a bicarbonate ion (HCO3-) and a hydrogen ion (H+). Because of the combined presence of a weak acid (H2CO3) and a conjugate base (HCO3-), any excess acid or base that comes into the system is neutralized. This keeps the pH balanced.